 
The exact value can vary widely, depending on material factors such as purity and phase as well as physical parameters such as temperature. Some approximate values of common ceramics are listed below (Haruta and Brunelle, except where noted). At pH values above the IEP, the predominate surface species is M-O -, while at pH values below the IEP, M-OH + species predominate. For these surfaces, present as colloids or larger particles in aqueous solution, the surface is generally assumed to be covered with surface hydroxyl species, M-OH (where M is a metal such as Al, Si, etc.). The isoelectric points (IEP) of metal oxide ceramics are used extensively in material science in various aqueous processing steps (synthesis, modification, etc.). This is also true for individual amino acids. If the protein is run with a buffer pH that is equal to the pI, it will not migrate at all. If the pH of the buffer is below the pI of the protein being run, the protein will migrate to the negative pole of the gel (positive charge is attracted to the negative pole). If the pH of the buffer is above the pI of the protein being run, the protein will migrate to the positive pole (negative charge is attracted to a positive pole). 
The pH of an electrophoretic gel is determined by the buffer used for that gel. This has implications for running electrophoretic gels (see Agarose gel electrophoresis). Above the pI they carry a net negative charge. Proteins can be separated according to their isoelectric point in a process known as isoelectric focusing.Īt a pH below the pI, proteins carry a net positive charge. However, a more exact treatment of this requires advanced acid/ base knowledge and calculations. Isoelectric focusing is also the first step in preforming 2-D gel polyacrylamide gel electrophoresis.įor an amino acid with only one amine and one carboxyl group, the pI can be calculated from the pKas of this molecule. 
This technique utilises a pH gradient to separate proteins. Proteins can be separated according to their isoelectric point (overall charge) on a polyacrylamide gel using a technique called isoelectric focusing. Above their pI they carry a net negative charge. At a pH below their pI, proteins carry a net positive charge. Amino acids which make up proteins may be positive, negative, neutral or polar in nature, and together give a protein its overall charge. Biological amphoteric molecules such as proteins contain both acidic and basic functional groups. Such molecules have minimum solubility in water or salt solutions at the pH which corresponds to their pI and are often seen to precipitate out of solution. A molecule's pI can affect its solubility at a certain pH. They are affected by pH of their surrounding environment and can become more positively or negatively charged due to the loss or gain of protons (H +). Amphoteric molecules called zwitterions contain both positive and negative charges depending on the functional groups present in the molecule. The isoelectric point (pI) is the pH at which a particular molecule or surface carries no net electrical charge. 3 Isoelectric Point versus Point of Zero Charge.2.1 Examples of isoelectric points for various materials.2 Isoelectric Point of Ceramic Materials.List of terms related to Isoelectric point 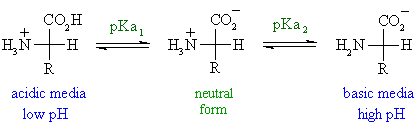
Risk calculators and risk factors for Isoelectric pointĬauses & Risk Factors for Isoelectric point US National Guidelines Clearinghouse on Isoelectric pointĭirections to Hospitals Treating Isoelectric point Ongoing Trials on Isoelectric point at Clinical Ĭlinical Trials on Isoelectric point at Google Most recent articles on Isoelectric pointĪrticles on Isoelectric point in N Eng J Med, Lancet, BMJĬochrane Collaboration on Isoelectric point 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
